I am text block. Click edit button to change this text. Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Company Profile
Founded in 2011 by CEO John Ruprai, Ennogen is a privately owned British company offering a range of medicines, medical devices, unlicensed medicines, and OTC products.
Since it was established, Ennogen has developed a differentiated portfolio of over 150 Market Authorisations, holds the ISO13485 accreditation and is the legal manufacturer and distributor of a range of medical devices.
Our commercial coverage extends across the entire UK mainline and shortline wholesale channels, private and NHS hospitals, community and retail pharmacies and Clinical Commissioning Groups.
Ennogen continues to establish itself as a leading choice for pharmacists and clinicians for numerous products. Our commitment is to provide quality and affordable pharmaceutical products to our local, national, and international clientele in an ethical, efficient and professional manner.
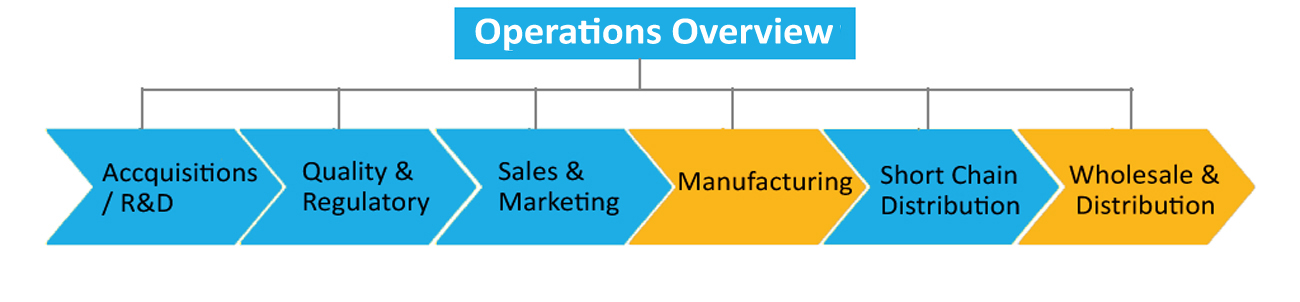
Ennogen Overview

Quality Policy
We will maintain a Quality management system to meet the requirements of regulatory standards: “The Rules governing medicinal products in the European Union” and the latest version of ISO 13485 as certified and as indicated on Ennogen site certificates.
Our key focus is to ensure that our medicines and medical devices do not place patients at risk due to inadequate safety, quality or efficacy
We strive to provide our customers, patients and healthcare professionals with quality products, services and support to a standard that exceeds our market competitors.
The Quality management system will be periodically reviewed for its effectiveness and suitability.
We set Quality Objectives at various levels and functions within the organisation and with our suppliers that are relevant to the quality of our products.
Through our Management Review Process, we will measure our performance against our quality objectives and identify opportunities for improvement.
We communicate the review of our quality performance internally with our employees.
We are committed to understanding and meeting our customer needs by reacting to developing products in consultation with our customers, customer feedback, patient needs and market changes.
Company Profile
Founded in 2011 by CEO John Ruprai, Ennogen is a privately owned British company offering a range of medicines, medical devices, unlicensed medicines, and OTC products.
Since it was established, Ennogen has developed a differentiated portfolio of over 150 Market Authorisations and now holds the ISO13485 accreditation and is the legal manufacturer and distributor of a range of medical devices.
Our commercial coverage extends across the entire UK mainline and shortline wholesale channels, private and NHS hospitals, community and retail pharmacies and Clinical Commissioning Groups.
Ennogen continues to establish itself as a leading choice for pharmacists and clinicians for numerous products. Our commitment is to provide quality and affordable pharmaceutical products to our local, national, and international clientele in an ethical, efficient and professional manner.
Quality Policy
We maintain a quality management system to meet the requirements of regulatory standards: “The Rules governing medicinal products in the European Union” and the latest version of ISO 13485 as certified and as indicated on Ennogen site certificates. Our key focus is to ensure that our medicines and medical devices do not place patients at risk due to inadequate safety, quality, or efficacy.
We strive to provide our customers, patients, and healthcare professionals with quality products, services, and support to a standard that exceeds our market competitors. The Quality management system will be periodically reviewed for its effectiveness and suitability.
We set Quality Objectives at various levels and functions within the organization and with our suppliers that are relevant to the quality of our products. Through our senior management review process, we will measure our performance against our quality objectives and identify opportunities for improvement. We communicate the review of our quality performance internally with our employees.
We are committed to understanding and meeting our customer needs by reacting to developing products in consultation with our customers, customer feedback, patient needs, and market changes.
Ennogen Overview










